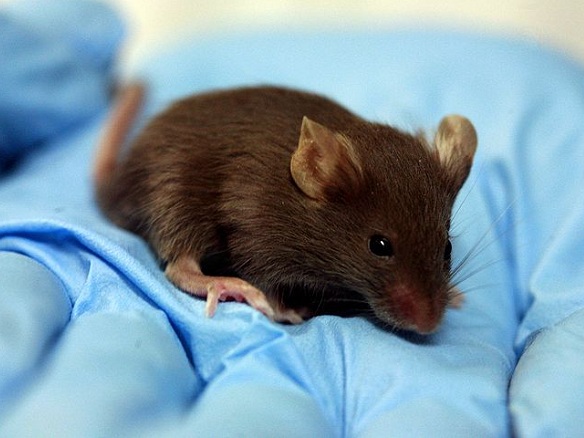
Researchers looking for genetic causes of disease have begun going through the whole mouse genome in a bid to understand human disorders. Results will hopefully help find new therapies which treat underlying causes and not just symptoms.
The aim is to knock out one gene at a time and look for physical changes. Some gene knock-outs are fatal and the mice do not survive, indicating the importance of the gene in early development. Mice which do survive are tested and any diseases or abnormalities which develop are linked back to the original genetic change.
Over the next ten years, 20,000 mouse mutants will be made and examined by 18 research centres around the world. The Medical Research Council’s Harwell centre in Oxfordshire is part of the project, called the International Mouse Phenotyping Consortium. MRC Harwell is set to breed and test around 10 percent of mice in the project . A significant number of the 50,000 mice who live at Harwell are being used, as 14 copies of each mutant mouse are made.
Researchers will build upon previous work from the EUMODIC project which acted as a pilot study and looked at fewer genes.

Each yellow box houses one type of mutant mouse. Thousands have been made to model human conditions.
The pilot project’s results have so far resulted in various mutations being linked to brain dysfunction, heart problems and diabetes. Scientists believe the importance lies in finding links between different gene mutations and not just linking one gene to a single disease.
“To some extent, it’s not so much findings we get from one mutation but more the collective information because ultimately genes do not work in isolation. They are all part of pathways and networks that make us, build us as we develop and make us work properly as an adult,” said Professor Steve Brown, Director of the Mammalian Genetics Unit at MRC Harwell.
As mammals, mice have similar body machinery to humans. They are also genetically similar to us, so genetic mutations which cause a particular abnormality in the mouse correlate to humans. This is the first time a study will systematically inspect the entire genome of a mammal. It will build on knowledge gained previously from unlikely organisms such as yeast, flies and worms and make findings more relevant to humans.
Eventually, researchers will be able to “put a tag on each gene and say that’s how that gene is involved in human disease and that is tremendously powerful because everything has a genetic component,” said Prof Brown. “We really feel that as we go from using hundreds to thousands of mice we will generate a rich database that everyone from a basic scientist to a clinician can use.”
The mutants
The genes are knocked out in an early stage of development using very sophisticated genetic engineering. Seven males and seven females for each mutation are made, so that there are enough mice to analyse to obtain results significant enough to draw conclusions from.

Newborn mice take 21 days to feed independently. Phenotyping tests begin when the mutants are 9 weeks old.
Each mutant mouse is left to grow as normal until at aged 9 weeks when the “phenotyping” tests begin. There are 20 different tests which investigate everything from movement to metabolism to chemicals in the blood. If an abnormality is found, a connection can be made between the altered gene and resultant observable presence, or phenotype, of disease. After the “in-life” tests, the mice are sacrificed and further investigations begin as specialists examine blood and organs in more detail.
All results are entered into a freely-accessible online database which lists all analysed genes and highlights where abnormalities have been found.
Future study will account for mouse ageing, Prof Brown said. “For pragmatic and logistical reasons at the moment we are not keeping a cohort on shelf to see what diseases develop later in life. As we know a lot of illnesses are disease of ageing so this is an important aspect to bring to programme in future. This project will produce a huge amount of interesting information, but there is always more to do.”